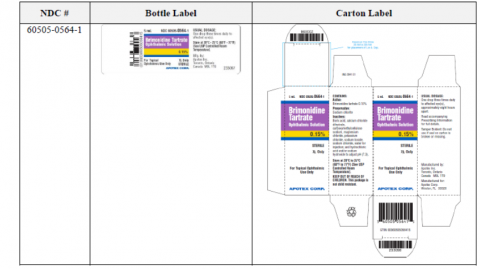
Apotex Corp. Issues Voluntary Nationwide Recall of Brimonidine Tartrate Ophthalmic Solution, 0.15% Due to Cracks that Have Developed in Some of the Units Caps of the Bottles | FDA

Bacteria in recalled eye drops linked to cases of vision loss, surgical removal of eyeballs | Recalls | abc12.com

Apotex Corp. Issues Voluntary Nationwide Recall of Brimonidine Tartrate Ophthalmic Solution, 0.15% due to cracks that have developed in some of the units caps of the bottles